Current Suspected Overdose Deaths in Delaware for 2024: Get Help Now!
Find school water testing results and additional resources
Attention Medicaid Participants: Eligibility Renewals Restarted April 1, 2023
Creutzfeldt-Jakob disease (CJD) is a rare, fatal brain disorder believed to be caused by a protein called a prion. A prion is a nonliving, self-replicating infectious agent made of protein. It is thought to arise from a change in shape of a prion protein. This abnormally-folded protein is then thought to cause other prion proteins to change shape. This process leads to the destruction of brain cells.
CJD is one of a family of related diseases found in many animal species, called Transmissible Spongiform Encephalopathies (TSEs). These include scrapie in sheep and Bovine Spongiform Encephalopathy (BSE) in cattle. BSE is also known as ‘Mad Cow disease.’
CJD is not related to Mad Cow disease.
|
CJD, the disease in humans, can take one of four forms including sporadic (sCJD), familial or inherited, acquired or iatrogenic and variant (vCJD).
|
Basic
|
Support is a key ingredient in coping with CJD. Support from those who have had similar experiences can be more helpful than any written information. The primary goal of the CJD Foundation is to provide support for CJD patients and their families.
World Health Organization (WHO) Guidelines for Transmissible Spongiform Encephalopathies (TSE), March 1999
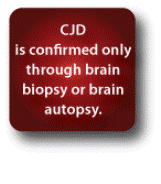
The NPDPSC was established in 1997 at the Division of Neuropathology of Case Western Reserve University and is sponsored by the American Association of Neuropathologists (AANP). The NPDPSC monitors the occurrence of prion disease through characterization of brain tissue pathology from clinically diagnosed cases of CJD. A confirmatory diagnosis of CJD can only be made through brain biopsy or brain autopsy. Autopsy provides the only method to fully classify the type of CJD (sporadic, familial, iatrogenic, or variant). The NPDPSC can help establish the diagnosis of prion disease by analyzing cerebrospinal fluid (CSF), blood, and brain tissue obtained either at biopsy or autopsy.
For questions regarding services offered by the NPDPSC or assistance in arranging an autopsy, please call the Center at (216-368-0587).
Although there have been no documented cases of a patient developing CJD from a transfusion of blood or plasma, all potential donors are screened to determine if they fall into a CJD risk group that has been determined by the Food and Drug Administration (FDA).
BBD defers all potential donors who may be at risk for CJD according to the following criteria:
NOTE: A person who has only one family member with CJD may be at increased risk for developing the disease. However, he or she is much less likely to develop CJD than a person who has two or more family members diagnosed with CJD.
For more information, contact Blood Bank of Delmarva Donor Advocate at (302)737-8405 (or 1-800-533-6957).
CJD became a reportable condition in Delaware in 2006: Regulations for the Control of Communicable and Other Disease Conditions
Purpose of Reporting and Surveillance:
Healthcare providers should contact the Delaware Division of Public Health (DPH), Bureau of Epidemiology to report any suspect case of CJD.
Office of Infectious Disease Epidemiology
24/7 Emergency Contact Number: 1-888-295-5156
Return to Epidemiology Home Page
![]() Please note: Some of the files available on this page are in Adobe PDF format which requires Adobe Acrobat Reader. A free copy of Adobe Acrobat Reader can be downloaded directly from Adobe . If you are using an assistive technology unable to read Adobe PDF, please either view the corresponding text only version (if available) or visit Adobe's Accessibility Tools page.
Please note: Some of the files available on this page are in Adobe PDF format which requires Adobe Acrobat Reader. A free copy of Adobe Acrobat Reader can be downloaded directly from Adobe . If you are using an assistive technology unable to read Adobe PDF, please either view the corresponding text only version (if available) or visit Adobe's Accessibility Tools page.